
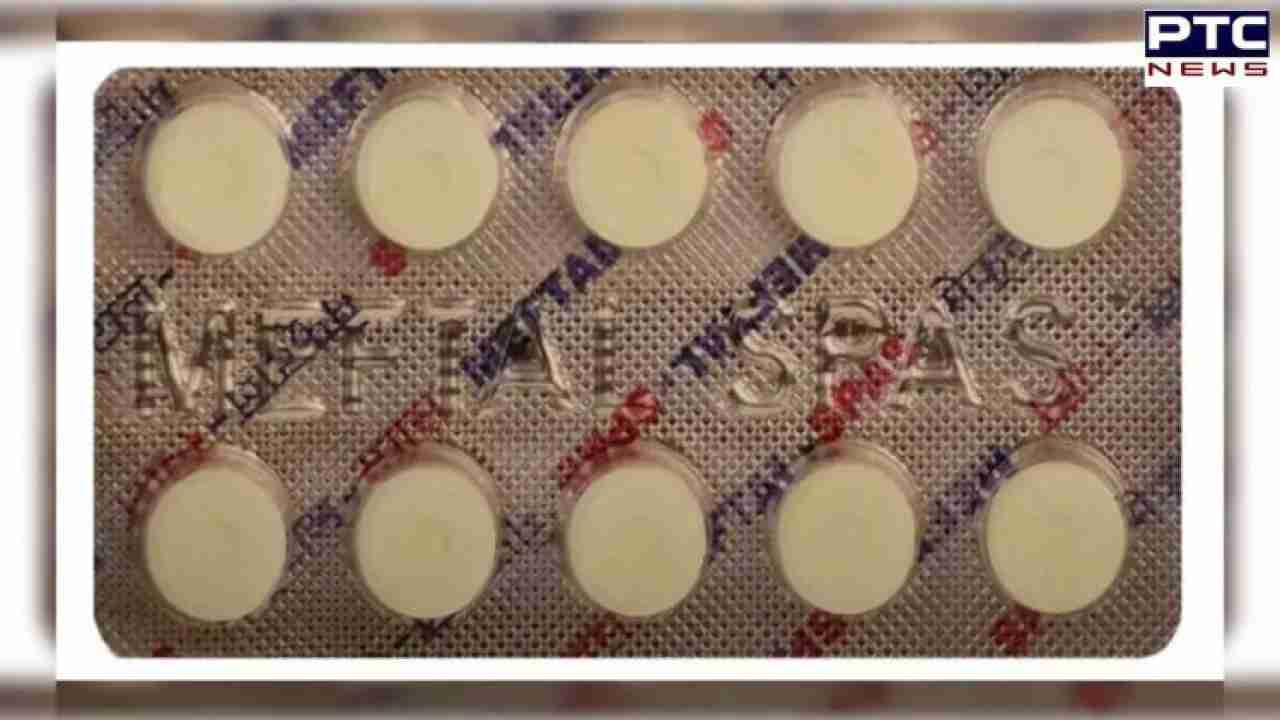
Is popping painkiller Meftal risky? Government issues safety alert
Meftal risky: The Indian Pharmacopoeia Commission (IPC) has raised concerns about the use of Meftal, a painkiller containing mefenamic acid, citing potential adverse reactions associated with the drug. Specifically, the commission has highlighted the risk of Drug Reactions with Eosinophilia and Systemic Symptoms (DRESS) syndrome linked to this medication.
Meftal, commonly prescribed for conditions like rheumatoid arthritis, osteoarthritis, dysmenorrhoea, mild to moderate pain, inflammation, fever, and dental pain, is under scrutiny due to this alert. DRESS syndrome is a severe allergic reaction that may manifest as skin rash, fever, and lymphadenopathy, typically emerging within two to eight weeks after taking the drug.
In a statement issued on November 30, the IPC emphasized the importance of close monitoring for potential adverse drug reactions associated with Meftal. Healthcare professionals and patients alike are strongly advised to be vigilant. The commission's release outlined precautionary measures, urging individuals to promptly report any suspected adverse reactions to the national coordination center of the Pharmacovigilance Programme of India (PvPI). Reporting can be done through the commission's website (www.ipc.gov.in), the ADR PvPI android mobile app, or by contacting the PvPI helpline at 1800-180-3024.
The IPC stresses the need for heightened vigilance, asking individuals to remain attentive to symptoms suggestive of DRESS syndrome. Should any such reactions occur, swift reporting to the national coordination center of the PvPI is encouraged.
- With inputs from agencies
