
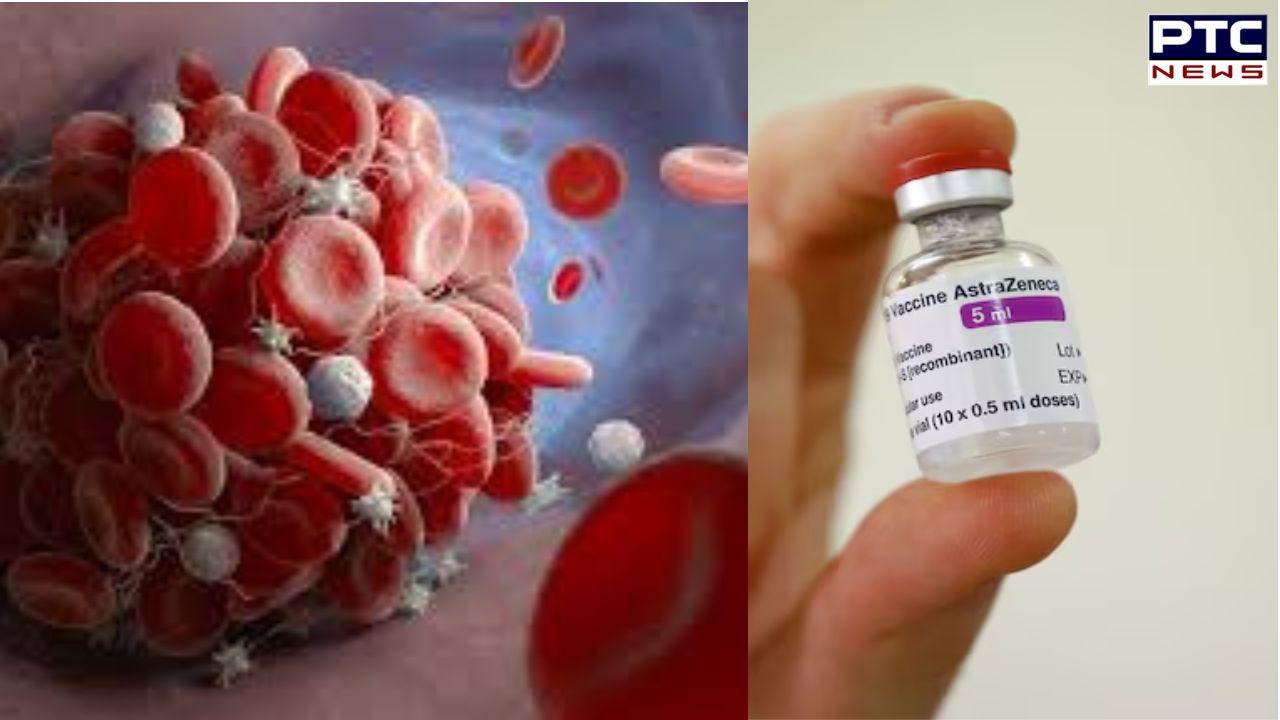
Received AstraZeneca's Covishield Vaccine? It's time for Thrombosis with Thrombocytopenia Syndrome testing | In Detail
PTC Web Desk: In a significant development, British pharmaceutical giant AstraZeneca has publicly acknowledged that its Covid-19 vaccine may lead to a rare side effect known as Thrombosis with Thrombocytopenia Syndrome (TTS). This admission comes on the heels of a lawsuit filed against the company, alleging serious harm and even deaths associated with the vaccine.
The lawsuit underscores growing concerns surrounding Covishield, a vaccine jointly developed by AstraZeneca and Oxford University and manufactured by the Serum Institute of India. Covishield was widely administered throughout India during the peak of the Covid-19 pandemic, raising questions about its safety profile.
Understanding Thrombosis with Thrombocytopenia Syndrome (TTS)
Thrombosis with Thrombocytopenia Syndrome (TTS) is a rare medical condition characterised by the formation of blood clots in unusual locations within the body, coupled with a decrease in the number of platelets in the bloodstream. Platelets play a crucial role in blood clotting, so a reduced platelet count can pose significant health risks. TTS has been observed in individuals who have received adenoviral vector Covid-19 vaccines, including Vaxzevria, Covishield (AstraZeneca), and the Johnson & Johnson/Janssen Covid-19 vaccine. This syndrome is believed to occur due to an immune response triggered by the vaccine, leading to the production of antibodies that target a specific protein involved in blood clotting.
Classification of TTS by CDC
TTS is categorised into two tiers by the Centres for Disease Control and Prevention (CDC), based on the severity of symptoms and associated risks:
Tier 1: This tier encompasses cases where rare blood clots occur in critical locations such as the brain or gastrointestinal tract, sometimes in conjunction with more typical clots in the legs or lungs. Individuals affected by Tier 1 TTS typically exhibit a low platelet count (below 150,000 per microliter). While a positive anti-PF4 ELISA test can aid in confirming the diagnosis, it may not always be necessary. Tier 1 cases are generally considered more severe and pose higher risks, with a higher prevalence observed among younger demographics.
Tier 2: In Tier 2 cases, people experience more common blood clots, such as those in the legs or lungs, accompanied by a low platelet count. Diagnosis of Tier 2 TTS requires a positive anti-PF4 ELISA test. While less severe than Tier 1 cases, Tier 2 TTS still presents significant health concerns and requires prompt medical attention.
TTS Symptoms
Symptoms of TTS can manifest in various ways, including severe headaches, abdominal pain, swelling in the legs, difficulty breathing, and cognitive impairment or seizures. Individuals who experience these symptoms following vaccination should seek immediate medical evaluation to rule out TTS and receive appropriate care.
- With inputs from agencies
