
PGI Chandigarh begins clinical trials of Oxford vaccine; three volunteers given first dose
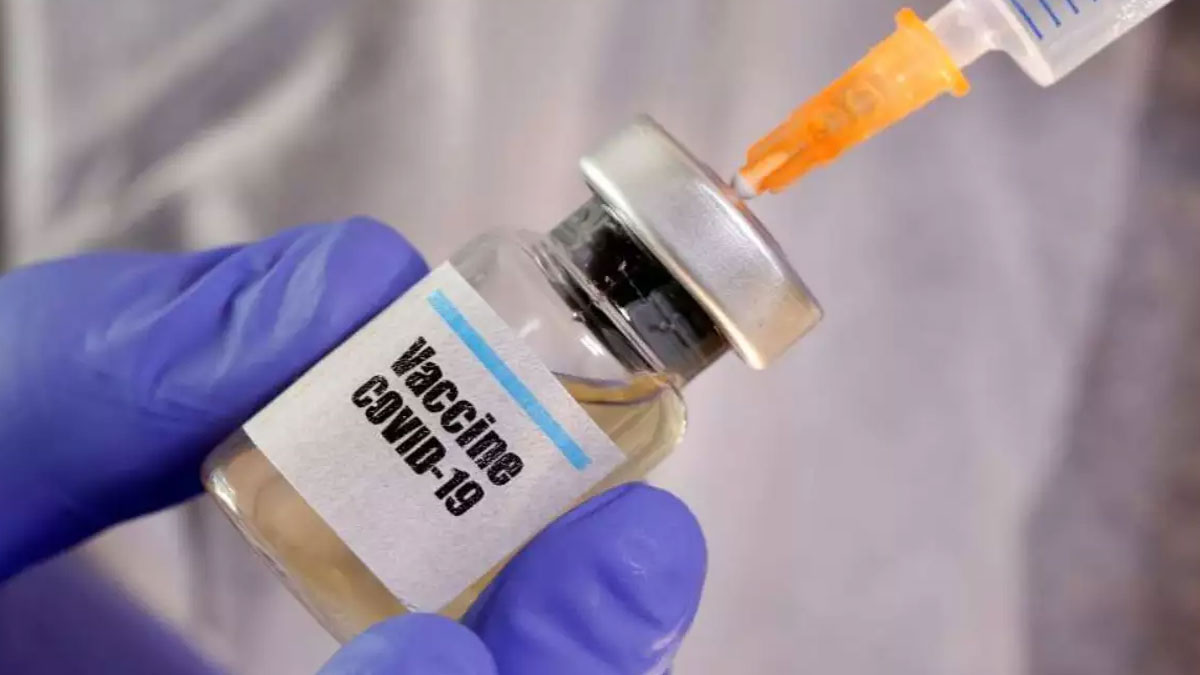
The Post Graduate Institute of Medical Education and Research (PGIMER), Chandigarh, has administered the first dose of Oxford Covid-19 vaccine named Covishield to three candidates on Friday, informed PGI Chandigarh Director, Prof. Jagat Ram.
 He said that two females and a male were given dosage of 0.5 ml.
"Two females aged 57 and 26 years and a 33-year-old male were administered the vaccine candidate at the institute," he stated.
Also Read | IAF's Rafale squadron to get its first woman fighter pilot
The PGIMER is one of the seventeen sites where phase II trials of the 'Covishield' vaccine developed by Oxford University and being mass-produced by Pune based Serum Institute of India (SII) is being conducted.
He said that two females and a male were given dosage of 0.5 ml.
"Two females aged 57 and 26 years and a 33-year-old male were administered the vaccine candidate at the institute," he stated.
Also Read | IAF's Rafale squadron to get its first woman fighter pilot
The PGIMER is one of the seventeen sites where phase II trials of the 'Covishield' vaccine developed by Oxford University and being mass-produced by Pune based Serum Institute of India (SII) is being conducted.
 According to the sources, the candidates will be monitored for 28 days after the 0.5 ml dose of vaccine and then the second dose will be given followed by monitoring of 28 days.
After that a blood sample will be taken and they will be observed for a span of six months.
According to the sources, the candidates will be monitored for 28 days after the 0.5 ml dose of vaccine and then the second dose will be given followed by monitoring of 28 days.
After that a blood sample will be taken and they will be observed for a span of six months.
 Besides, the candidates have been asked to note any development of symptoms like a fever in their personal diary. 18 candidates have been found eligible till now to undergo the trial. The institute plans to take 100 candidates into the phase II study while it has made a commitment for 253 candidates to Indian Council of Medical Research (ICMR).
Also Read | Trump admin proposes fixed time limit for different visas in US
The trials started after the institute received safety approval pending from the Data Safety and Monitoring Board (DSMB) for the first 100 candidates selected to undergo the trials last week.
Besides, the candidates have been asked to note any development of symptoms like a fever in their personal diary. 18 candidates have been found eligible till now to undergo the trial. The institute plans to take 100 candidates into the phase II study while it has made a commitment for 253 candidates to Indian Council of Medical Research (ICMR).
Also Read | Trump admin proposes fixed time limit for different visas in US
The trials started after the institute received safety approval pending from the Data Safety and Monitoring Board (DSMB) for the first 100 candidates selected to undergo the trials last week.
 The criteria fulfilling the eligibility prerequisites for the study included the minimum age of 18, no underlying medical condition and no prior Covid-19 infection. Besides, family members of the candidates are also required to be COVID negative.
-PTC News
The criteria fulfilling the eligibility prerequisites for the study included the minimum age of 18, no underlying medical condition and no prior Covid-19 infection. Besides, family members of the candidates are also required to be COVID negative.
-PTC News
